Hoechst 33258 trihydrochloride [23491-45-4]
Référence NB-64-27871-25mg
Conditionnement : 25mg
Marque : Neo Biotech
Hoechst 33258 trihydrochloride (Synonyms: Pibenzimol, Hoechst 33258, HOE 33258, H 33258 trihydrochloride, bi...)
Catalog No. T4057 Copy Product Info
Purity: 99.47%
Hoechst 33258 trihydrochloride (Bisbenzimide) is a benzimidazole anti-filarial agent. it is fluorescent when it binds to certain nucleotides in DNA, thus providing a tool for the study of DNA replication. it also interferes with mitosis.
Hoechst 33258 trihydrochloride
Copy Product InfoSynonyms Pibenzimol, Hoechst 33258, HOE 33258, H 33258 trihydrochloride, bisBenzimide H 33258 trihydrochloride, Bisbenzimide
Hoechst 33258 trihydrochloride (Bisbenzimide) is a benzimidazole anti-filarial agent. it is fluorescent when it binds to certain nucleotides in DNA, thus providing a tool for the study of DNA replication. it also interferes with mitosis.
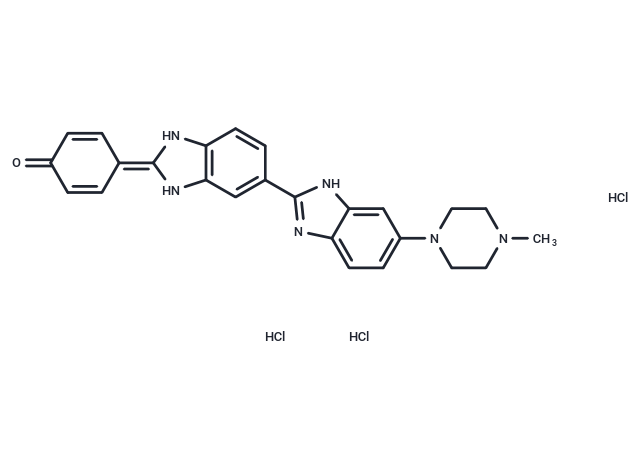
Cas No. 23491-45-4
Select Batch
Purity:99.47%
Appearance:Solid
Color:White to Yellow
Contact us for more batch information
Product Introduction
Hoechst 33258 trihydrochloride AI Summary
Hoechst 33258 trihydrochloride exhibits multifunctional bioactivities, including binding affinities to nucleic acids and notable antibacterial properties. It demonstrates a higher binding affinity to Poly (dA-dT) with a C50 value of 0.2 µM, compared to Poly (dG-dC) with a C50 value of 1.4 µM, as evidenced by ethidium displacement assays. The compound also inhibits the binding of ethidium bromide to RNA and binds to the poly(rA).(rU) RNA duplex, causing changes in melting temperature and hypochromicity. Furthermore, Hoechst 33258 trihydrochloride shows antibacterial activity against multiple strains such as Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, and Pseudomonas aeruginosa, with MIC values ranging from 8.0 to 16.0 µg/mL. It is minimally cytotoxic to human DU145 cells (IC50 > 10,000 nM). Its antiproliferative efficacy is observed in human HeLa cells (IC50 = 23,520 nM), MCF7 cells (IC50 = 48,100 nM), and KG-1 cells (IC50 = 42,000 nM) after 48 hours via MTT assays. Moreover, Hoechst 33258 trihydrochloride binds biotinylated pre-miRNA-21 with a Kd value of 100 nM as shown by SPR analysis and inhibits the activities of Topoisomerase 1 and Topoisomerase 2 each with an IC50 value of 30,000 nM..
Note: Summary generated by AI. Data source: ChEMBL 
Bioactivity
Chemical Properties
Storage & Solubility Information
| Description | Hoechst 33258 trihydrochloride (Bisbenzimide) is a benzimidazole anti-filarial agent. it is fluorescent when it binds to certain nucleotides in DNA, thus providing a tool for the study of DNA replication. it also interferes with mitosis. |
| Targets&IC50 | HeLa cells:51.31±4.56 μM (IC50), U-937 cells:15.42±2.16 μM (IC50), HL-60 cells:32.43±3.27 μM (IC50) |
| Cell Research | Solution preparation a. Preparation of stock solution: Dissolve 10 mg in 5 mL DMSO; (It is recommended to store at -20 ℃ or -80 ℃ in the dark after aliquoting) b. Preparation of Hoechst working solution Dilute the stock solution and dissolve it in serum-free cell culture medium or PBS to obtain a Hoechst working solution with a final concentration of 10 μg/mL; (Select the appropriate working solution concentration according to experimental requirements and prepare it for immediate use) c. Cell staining 1. Suspended cells (6-well plate) a. Centrifuge at 1000 g for 3-5 minutes at 4℃, and then discard the supernatant. Wash twice with PBS for 5 minutes each. The cell density is 1×106/mL; b. Add 1 mL of working solution and incubate at room temperature for 3-10 minutes; c. Centrifuge at 400 g for 3-4 minutes at 4℃, and discard the supernatant; d. Wash twice with PBS, 5 minutes each time; e. Resuspend cells with serum-free cell culture medium or PBS. Observe with fluorescence microscope or flow cytometer. 2. Adherent cells a. Culture adherent cells on sterile coverslips; b. Remove coverslips from culture medium and aspirate excess culture medium; c. Add 100 μL working solution, shake gently to completely cover cells, and incubate at room temperature for 3-10 minutes; d. Wash twice with culture medium, 5 minutes each time. Observe with fluorescence microscope or flow cytometer. The above information is based on published literature. Experimental procedures should be appropriately modified to meet specific research demands. |
| Synonyms | Pibenzimol, Hoechst 33258, HOE 33258, H 33258 trihydrochloride, bisBenzimide H 33258 trihydrochloride, Bisbenzimide |
| Molecular Weight | 533.9 |
| Formula | C25H24N6O·3HCl |
| Cas No. | 23491-45-4 |
| Smiles | CN1CCN(c(cc2)cc3c2nc(c4cc5c(N/C(N5)=C6C=CC(C=C\6)=O)cc4)[nH]3)CC1.Cl.Cl.Cl |
| Relative Density. | 1.5099 g/cm3 (Estimated) |
| Storage | Shipping with blue ice/Shipping at ambient temperature. | ||||||||||||||||||||
| Solubility Information | DMSO: 6.58 mg/mL (12.32 mM), Sonication is recommended.  | ||||||||||||||||||||
Solution Preparation Table | |||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | |||||||||||||||||||||



